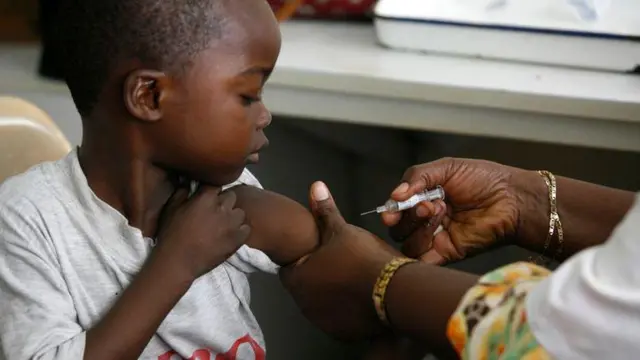
The first malaria treatment specifically formulated for babies and very young children has been approved and is set to be rolled out across African countries in the coming weeks.
Known as Coartem Baby (or Riamet Baby in some regions), the drug was developed by Novartis in partnership with the Medicines for Malaria Venture (MMV), a Swiss-based non-profit supported by governments including the UK, Switzerland, and the Netherlands, as well as organizations such as the World Bank and Rockefeller Foundation.
Until now, babies weighing less than 4.5 kg (around 10 pounds) have been treated with malaria drugs designed for older children — a practice that carries a risk of overdose, as infants’ developing livers process medication differently.
This gap in treatment options has contributed to ongoing health risks in this vulnerable age group. According to 2023 figures, malaria caused approximately 597,000 deaths, nearly all in Africa, with about 75% occurring in children under five.
The new medicine has been approved by Swiss authorities and is expected to be made available at near cost in countries hardest hit by malaria.
Novartis and MMV hope the drug will significantly reduce malaria-related deaths among the youngest children by providing a safer, tailored treatment option.